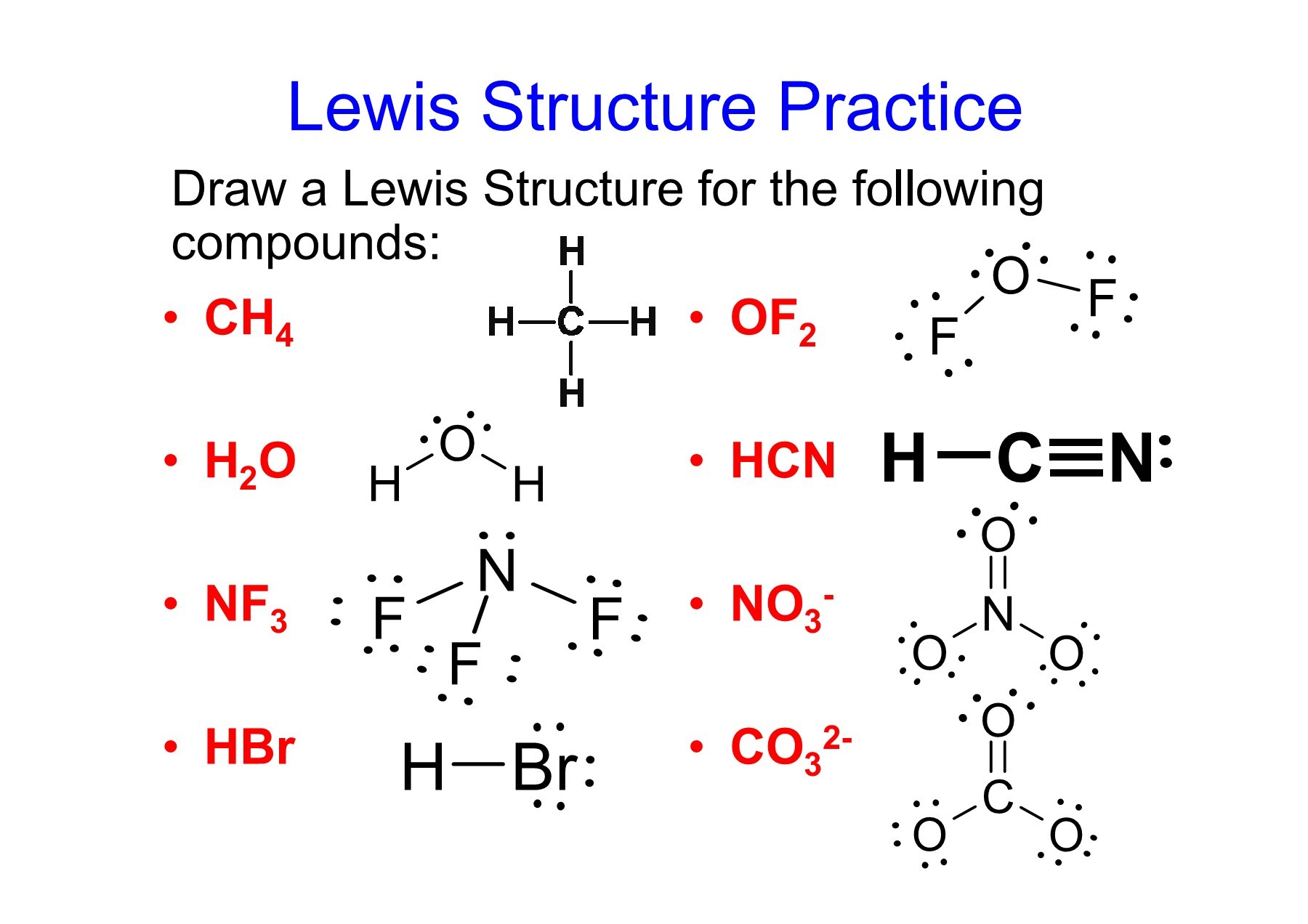
Understanding the Lewis Structure of HBr is profound for anyone examine alchemy, as it provides insights into the soldering and properties of this mere yet important compound. Hydrogen platitude (HBr) is a diatomic atom consist of one hydrogen atom and one br atom. By examining its Lewis construction, we can determine the arrangement of electrons, the type of bond make, and the overall stability of the corpuscle.
What is a Lewis Structure?
A Lewis structure, also known as a Lewis dot diagram, is a diagrammatic representation of the valence electrons in a mote. It help visualize the bonding between speck and the lone span of negatron. The structure is named after Gilbert N. Lewis, who acquaint the concept in 1916. Lewis construction are essential for understanding the chemic behavior of molecule, include their reactivity and constancy.
Steps to Draw the Lewis Structure of HBr
Pull the Lewis construction of HBr involves several stairs. Hither's a elaborate guide:
Step 1: Determine the Total Number of Valence Electrons
The 1st measure is to count the total act of valency electrons in the speck. Hydrogen has one valency electron, and br has seven valence electrons. Thus, the entire number of valence negatron in HBr is:
1 (from H) + 7 (from Br) = 8 valency electrons
Step 2: Identify the Central Atom
In the case of HBr, br is the central atom because it is more negative than hydrogen. The central particle is typically the one with the high negativity or the one that can constitute more bond.
Step 3: Arrange the Atoms
Place the hydrogen atom and the br atom next to each other. Since HBr is a diatomic molecule, the agreement is straightforward:
H - Br
Step 4: Form the Bond
Use two electron to form a single covalent alliance between the hydrogen and br speck. This alliance represents the shared duet of electrons:
H: Br
Step 5: Distribute the Remaining Electrons
After spring the bond, distribute the remaining six valency electron around the bromine particle. Bromine needs eight electron to complete its viii, so it will have three lonely pairs of electron:
H: Br:
H: Br:
H: Br:
Step 6: Verify the Octet Rule
Ensure if both atoms have achieve a stable negatron configuration. Hydrogen has two electron (one from the bond and one from its valence negatron), and bromine has eight electrons (two from the alliance and six from its lone pairs). This satisfy the eight formula for br and the duet rule for hydrogen.
📝 Note: The octet rule state that atom lean to spring alliance to attain eight valence electrons, while the span prescript applies to hydrogen, which just needs two electrons to be stable.
Properties of HBr
The Lewis construction of HBr provides valuable info about its property. Some key property include:
- Polarity: HBr is a diametrical corpuscle because br is more electronegative than hydrogen. This results in a dipole instant, create HBr a diametrical particle.
- Alliance Character: The alliance between hydrogen and bromine is a single covalent alliance, formed by the sharing of two electrons.
- Molecular Form: HBr has a analogue molecular contour, which is distinctive for diatomic particle.
- Boiling and Melting Points: HBr has a boiling point of -66.8°C and a melting point of -86.9°C, indicating it is a gas at way temperature.
Applications of HBr
Hydrogen platitude has various crucial applications in various industries:
- Chemical Deduction: HBr is expend in the deduction of organic compounds, particularly in the product of brominated compound.
- Pharmaceuticals: It is used in the industry of sure pharmaceuticals and drug.
- Industrial Processes: HBr is use in various industrial process, include the production of flaming retardants and other chemical production.
Safety Considerations
Handling HBr command deliberate attention to safety due to its corrosive and toxic nature. Some refuge considerations include:
- Personal Protective Equipment (PPE): Use appropriate PPE, include gloves, safety glasses, and lab coats, when handling HBr.
- Airing: Ensure proper airing to obviate inhalation of HBr fumes, which can be harmful.
- Storage: Store HBr in a cool, dry place aside from uncongenial substances.
📝 Note: Always postdate safety protocol and guidelines when handling HBr to prevent fortuity and health risks.
Comparing HBr with Other Halogen Compounds
To better interpret the Lewis structure of HBr, it is helpful to liken it with other halogen compounds, such as HF, HCl, HBr, and HI. Hither is a comparing table:
| Compound | Cardinal Atom | Number of Valence Electrons | Bond Type | Polarity |
|---|---|---|---|---|
| HF | F | 8 | Single covalent | Polar |
| HCl | Cl | 8 | Single covalent | Polar |
| HBr | Br | 8 | Single covalent | Polar |
| HI | I | 8 | Single covalent | Polar |
As seen in the table, all these compound have a single covalent alliance and are opposite due to the electronegativity departure between hydrogen and the halogen atoms. The Lewis construction of HBr follows the same rule as these other halogen compounds.
Conclusion
The Lewis structure of HBr is a primal concept in chemistry that help us translate the soldering and properties of this important compound. By following the steps to describe the Lewis construction, we can project the agreement of electron and the type of alliance formed. The Lewis structure of HBr reveals that it is a polar molecule with a individual covalent bond, and it supply brainwave into its chemic behavior and applications. Understanding the Lewis construction of HBr is essential for anyone analyze chemistry, as it forms the base for more complex chemical concepts and reactions.
Related Footing:
- formal complaint of hbr
- hbr dot and cross diagram
- is hbr linear
- hbr molecular orbital diagram
- hbr molecular geometry
- hydrobromic battery-acid lewis construction