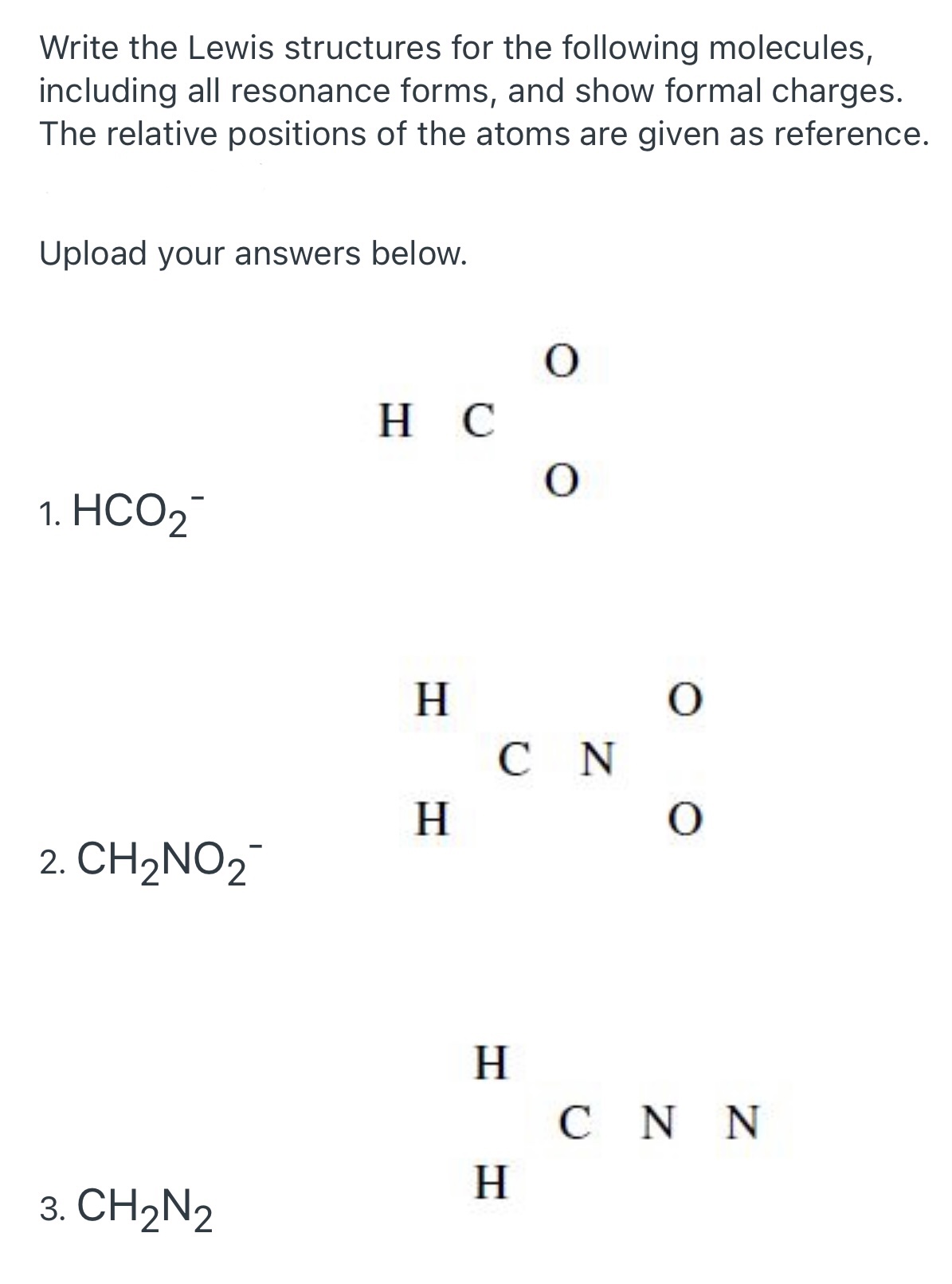
Understanding the Nof Lewis Structure is profound for anyone dig into the reality of alchemy. The Lewis structure, named after Gilbert N. Lewis, is a diagrammatical representation of the valence negatron in a molecule. It aid chemist visualize the bonding between mote and predict the molecular geometry. This construction is particularly useful for understanding the properties and demeanor of particle, do it an essential tool in both academic and industrial setting.
What is a Lewis Structure?
A Lewis construction, also know as an negatron dot structure, is a elementary way to symbolise the valency electrons of atom within a molecule. It apply transportation to represent electron and lines to symbolize covalent bond. The construction facilitate in understand the distribution of electron around atoms, which is essential for presage molecular properties.
Importance of Lewis Structures
Lewis structures are critical for respective reasons:
- Visual Representation: They provide a open visual representation of how atoms are stick within a corpuscle.
- Predicting Molecular Geometry: By understanding the Lewis structure, one can predict the chassis of the molecule, which is indispensable for understand its reactivity and properties.
- Electron Distribution: They aid in read the distribution of electrons, which is crucial for forebode the reactivity of molecules.
- Bonding Patterns: Lewis structures unwrap the type of alliance (individual, three-fold, ternary) and the front of lone couplet, which are crucial for understanding molecular behavior.
Steps to Draw a Lewis Structure
Delineate a Lewis structure imply several steps. Hither is a detailed guide:
- Count the Total Number of Valence Electrons: Determine the routine of valence negatron for each atom in the molecule and sum them up. for instance, in a h2o molecule (H 2 O), oxygen has 6 valence electrons, and each hydrogen has 1 valence electron, giving a total of 8 valence electrons.
- Determine the Central Atom: Place the central atom, which is usually the least negative speck. In H 2 O, oxygen is the central atom.
- Relate the Atoms with Single Bonds: Use single alliance to connect the central speck to the surrounding atoms. For H 2 O, this means connecting the oxygen atom to each hydrogen atom with a single bond.
- Distribute the Remaining Negatron: Place the stay valency negatron around the particle to finish their octonary (or duets for hydrogen). In H 2 O, after placing the single bonds, there are 4 electrons left, which are placed as two lone pairs on the oxygen atom.
- Check for Formal Charges: Calculate the formal charge on each particle to see the structure is stable. The formal complaint is compute as the number of valency electrons in the costless corpuscle minus the number of negatron depute to the particle in the Lewis construction.
📝 Tone: The octet convention province that atoms tend to have eight electrons in their valency shield to attain stability. Still, there are exception, such as corpuscle with expanded octets (e.g., SF 6 ) or molecules with incomplete octets (e.g., BF3 ).
Examples of Lewis Structures
Let's look at a few examples to exemplify the procedure of trace Lewis structure.
Water (H 2 O)
For the h2o molecule:
- Total valence negatron: 8 (6 from O + 1 from each H)
- Key corpuscle: Oxygen (O)
- Single bonds: O-H and O-H
- Continue electrons: 4 (put as two lone pairs on O)
- Formal charges: 0 on all corpuscle
Carbon Dioxide (CO 2 )
For the carbon dioxide molecule:
- Entire valence negatron: 16 (4 from C + 6 from each O)
- Key atom: Carbon ©
- Individual bond: C-O and C-O
- Remaining electron: 12 (order as double bonds between C and each O)
- Formal charges: 0 on all mote
Ammonia (NH 3 )
For the ammonia molecule:
- Total valence negatron: 8 (5 from N + 1 from each H)
- Central atom: Nitrogen (N)
- Single bonds: N-H, N-H, and N-H
- Remaining negatron: 2 (set as a lone couple on N)
- Formal charges: 0 on all corpuscle
Common Mistakes in Drawing Lewis Structures
While describe Lewis structures, it's common to make a few error. Hither are some to avoid:
- Incorrect Valence Electron Count: Ensure you enumerate the correct turn of valence electrons for each molecule.
- Incorrect Central Atom: Take the least electronegative atom as the cardinal atom.
- Improper Bonding: Shuffle sure to use the right turn of bond and lone pairs to fill the octet rule.
- Ignoring Formal Charges: Always check for formal charge to ensure the structure is stable.
Advanced Concepts in Lewis Structures
Beyond the basics, there are advanced conception that enhance the understanding of Lewis construction.
Resonance Structures
Resonance construction hap when a speck can be typify by multiple Lewis structures that differ solely in the view of electrons. for instance, the ozone molecule (O 3 ) has two resonance structures:

Expanded Octets
Some speck, peculiarly those in the third period and beyond, can have more than eight valence electrons. This is known as an expand octad. for illustration, sulphur in sulfur hexafluoride (SF 6 ) has 12 valence electrons.
Formal Charge Calculation
Formal charge is calculated using the recipe:
Formal Charge = (Number of valence electron in costless particle) - (Number of lone pair electrons + 1 ⁄2 Number of bond electrons)
for instance, in the nitrate ion (NO 3- ), the formal charge on the nitrogen atom is 0, while the formal charge on each oxygen atom is -1.
Applications of Lewis Structures
Lewis construction have legion applications in chemistry:
- Predicting Molecular Geometry: By understanding the Lewis structure, one can predict the bod of the molecule utilize the Valence Shell Electron Pair Repulsion (VSEPR) theory.
- Understanding Reactivity: Lewis structures help in understanding the reactivity of molecule by showing the distribution of negatron and the front of lone pairs.
- Contrive New Compounds: Apothecary use Lewis structure to contrive new compound with desired place by fudge the bonding and electron distribution.
- Analyzing Chemical Reactions: Lewis construction are used to analyze chemic response by showing the motility of electrons and the formation of new bonds.
Lewis Structures and Molecular Geometry
Lewis structures are intimately related to molecular geometry. The VSEPR hypothesis utilize the Lewis structure to predict the flesh of a molecule based on the repulsion between electron pairs. Here is a table showing the relationship between the number of electron dyad and the molecular geometry:
| Number of Electron Pairs | Molecular Geometry |
|---|---|
| 2 | Linear |
| 3 | Trigonal Planar |
| 4 | Tetrahedral |
| 5 | Trigonal Bipyramidal |
| 6 | Octahedral |
for instance, in the ammonia molecule (NH 3 ), the nitrogen atom has three bonding pairs and one lone pair, resulting in a trigonal pyramidal geometry.
Lewis Structures and Chemical Bonding
Lewis construction provide insights into the types of chemical bonds present in a molecule. There are three main character of bond:
- Single Bonds: Shared brace of electrons (e.g., H-H in H 2 )
- Double Bond: Two partake pairs of negatron (e.g., C=C in C 2 H4 )
- Three-fold Bonds: Three shared pairs of electrons (e.g., C≡C in C 2 H2 )
Interpret these alliance is crucial for portend the properties and reactivity of molecule. for instance, corpuscle with dual or triple alliance are oftentimes more reactive than those with individual bonds.
Lewis structures also supporter in understanding the concept of polar and nonpolar bonds. A opposite alliance occur when there is an inadequate distribution of electron between two atoms, ensue in a dipole instant. for representative, in the water atom (H 2 O), the O-H bonds are polar due to the higher electronegativity of oxygen.
In contrast, a nonpolar alliance occurs when there is an adequate distribution of electrons between two corpuscle. for instance, in the hydrogen speck (H 2 ), the H-H bond is nonpolar because both hydrogen atoms have the same electronegativity.
Lewis structures are a fundamental tool in alchemy, supply a clear and concise way to symbolize the soldering and negatron distribution in mote. By understanding how to draw and interpret Lewis structures, chemists can gain worthful perceptivity into the properties and conduct of molecules. This cognition is crucial for betoken molecular geometry, translate reactivity, designing new compounds, and analyzing chemical reaction.
Lewis structures are not just limited to simple molecules; they can also be applied to more complex systems, such as ions and polyatomic corpuscle. for instance, the sulphate ion (SO 42- ) has a central sulfur atom surrounded by four oxygen atoms, with resonance structures contributing to its stability.
to sum, the Nof Lewis Structure is a knock-down tool in the pharmacist's arsenal, offering a visual and nonrational way to understand the electronic structure of molecules. By overcome the art of reap Lewis construction, one can unlock a deeper savvy of chemical soldering, molecular geometry, and reactivity. This cognition is not simply crucial for donnish purposes but also has virtual applications in respective battleground, from pharmaceutical to textile science. Whether you are a scholar, a researcher, or a professional in the chemical industry, a solid compass of Lewis structures is indispensable for navigating the complex creation of chemistry.
Related Terms:
- nitrosyl lewis structure
- primary lewis construction of nof
- lewis construction for fno
- lewis dot structure for nof
- nof molecular geometry
- nof resonance construction