In the realm of electrochemical measurements, the truth and reliability of data are paramount. One of the critical components that guarantee precise measuring is the R501 Reference Electrode. This electrode plays a polar role in assorted electrochemical application, including erosion studies, battery enquiry, and environmental monitoring. Understanding the R501 Reference Electrode and its applications can significantly raise the lineament of electrochemical experiment and analyses.
Understanding the R501 Reference Electrode
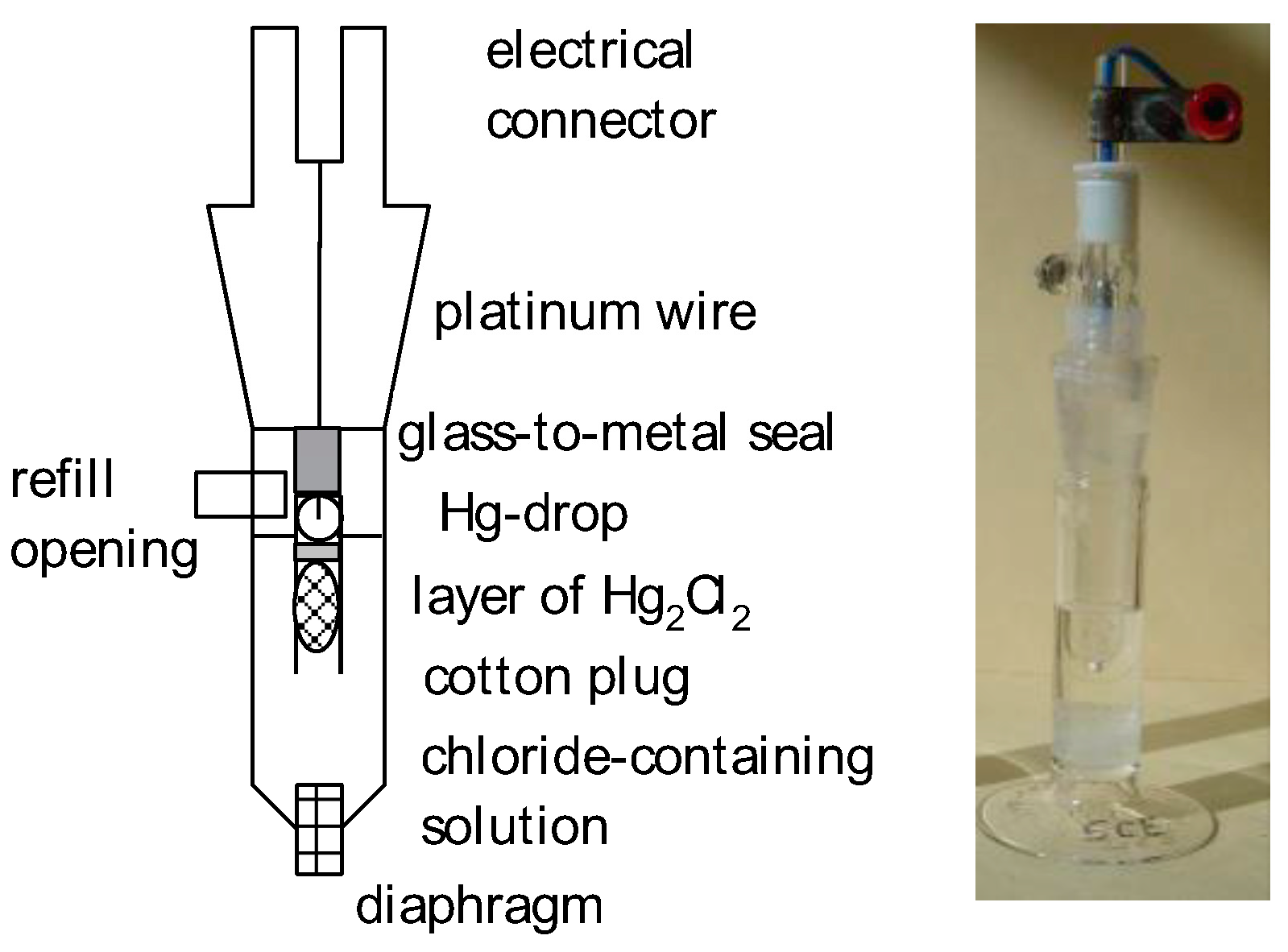
The R501 Reference Electrode is a type of reference electrode commonly used in electrochemical system. It provides a stable and known potentiality against which the potential of a working electrode can be measured. This constancy is essential for obtaining accurate and consistent answer in electrochemical experimentation.
The R501 Reference Electrode typically dwell of a silver/silver chloride (Ag/AgCl) electrode immerse in a potassium chloride (KCl) result. The Ag/AgCl electrode is known for its low drift and high stability, make it an idealistic option for reference electrode. The KCl resolution helps maintain a ceaseless ionic strength, further enhancing the constancy of the electrode.
Applications of the R501 Reference Electrode
The R501 Reference Electrode finds application in a blanket range of electrochemical report. Some of the key region where this electrode is commonly used include:
- Erosion Study: In corrosion research, the R501 Reference Electrode is employ to measure the corrosion potency of metal. This help in read the corroding conduct of cloth and evolve strategies to extenuate corroding.
- Battery Research: In battery research, the R501 Reference Electrode is used to analyse the electrochemical holding of battery materials. It help in characterizing the performance of batteries and developing new battery technology.
- Environmental Monitoring: The R501 Reference Electrode is also used in environmental monitoring to measure the density of pollutants in h2o. It helps in assessing the calibre of h2o and developing strategy to reduce befoulment.
Setting Up the R501 Reference Electrode
Setting up the R501 Reference Electrode involves several steps to ensure accurate and authentic mensuration. Here is a step-by-step guidebook to setting up the electrode:
- Ready the Electrode: Ensure that the R501 Reference Electrode is unclouded and free from any contaminant. Rinse the electrode with deionized h2o and dry it thoroughly.
- Fill the Electrode with KCl Solution: Filling the electrode with a concentrated KCl resolution. This solution helps preserve a constant ionic strength and enhances the constancy of the electrode.
- Join the Electrode: Connect the R501 Reference Electrode to the electrochemical scheme using appropriate line and connexion. Ensure that the connective are secure and free from any loose wires.
- Calibrate the Electrode: Fine-tune the electrode using a standard solution with a cognize voltage. This step ensures that the electrode is render precise measurement.
- Immerse the Electrode: Immerse the R501 Reference Electrode in the electrolyte solution of the electrochemical cell. Ensure that the electrode is fully overwhelm and that there are no air guggle around it.
🔍 Note: Always handle the R501 Reference Electrode with forethought to forefend any scathe. Ensure that the electrode is stored in a clear and dry surroundings when not in use.
Maintaining the R501 Reference Electrode
Maintaining the R501 Reference Electrode is all-important for ascertain its longevity and accuracy. Hither are some pourboire for keep the electrode:
- Regular Cleaning: Clean the electrode regularly to remove any contaminant that may touch its execution. Wash the electrode with deionized h2o and dry it exhaustively.
- Storage: Fund the electrode in a saturated KCl solution when not in use. This aid maintain the constancy of the electrode and prevents it from dry out.
- Calibration: Graduate the electrode periodically to check that it is providing precise measuring. Use a standard result with a known potency for calibration.
- Inspection: Inspect the electrode regularly for any signs of damage or wear. Replace the electrode if it prove any signs of deterioration.
🔍 Note: Avoid exposing the R501 Reference Electrode to extreme temperature or harsh chemicals, as this can damage the electrode and affect its execution.
Common Issues and Troubleshooting
Despite its dependability, the R501 Reference Electrode can sometimes meet issues that involve its execution. Here are some common issues and trouble-shoot steer:
| Number | Possible Reason | Solution |
|---|---|---|
| Inaccurate Measurements | Contamination or improper calibration | Clean the electrode and recalibrate it habituate a standard solution. |
| High Impetus | Depletion of KCl result or exposure to extreme temperatures | Replenish the electrode with tonic KCl solution and store it in a controlled environment. |
| Electrode Damage | Physical damage or exposure to coarse chemicals | Scrutinize the electrode for damage and replace it if necessary. |
🔍 Tone: Veritable upkeep and proper manipulation can preclude many of these topic and assure the longevity of the R501 Reference Electrode.
Advanced Applications of the R501 Reference Electrode
The R501 Reference Electrode is not restrict to basic electrochemical measurements. It also finds applications in advanced research country, such as:
- Electrochemical Impedance Spectroscopy (EIS): EIS is a powerful proficiency used to study the electrochemical holding of material. The R501 Reference Electrode is expend as a acknowledgment in EIS measurement to provide accurate and consistent results.
- Cyclic Voltammetry: Cyclic voltammetry is a technique expend to analyze the redox behavior of fabric. The R501 Reference Electrode is habituate as a reference in cyclic voltammetry to quantify the potential of the working electrode accurately.
- Potentiometric Titrations: Potentiometric titration are used to influence the density of ions in a solution. The R501 Reference Electrode is utilize as a mention in potentiometric titrations to render accurate measurements of the potential.
These advance applications foreground the versatility of the R501 Reference Electrode and its importance in respective electrochemical studies.

Future Trends in Reference Electrode Technology
The field of cite electrode technology is continually evolving, with new advancements aimed at better accuracy, stability, and strength. Some of the futurity swerve in acknowledgment electrode technology include:
- Miniaturization: The development of miniaturized credit electrodes that can be used in microelectrochemical systems. These electrodes are idealistic for applications where infinite is limited.
- Innovative Stuff: The use of advanced cloth, such as graphene and carbon nanotubes, to enhance the constancy and performance of reference electrode.
- Wireless Connectivity: The consolidation of tuner connectivity in reference electrodes to enable real-time monitoring and data collection.
These trends are expected to further enhance the potentiality of the R501 Reference Electrode and expand its covering in assorted field.
In drumhead, the R501 Reference Electrode is a important factor in electrochemical measurements, providing stable and accurate likely references. Its applications range from basic electrochemical work to advance research areas, making it an essential tool for scientists and engineers. Proper apparatus, upkeep, and troubleshoot are all-important for ensuring the reliability and seniority of the electrode. As reference electrode engineering keep to evolve, the R501 Reference Electrode will remain a cornerstone in the field of electrochemical measurement, driving founding and discovery in diverse scientific and industrial applications.