Xenon difluoride (XeF2) is a fascinating compound in the region of alchemy, particularly known for its unequaled properties and construction. One of the most intriguing aspects of XeF2 is its Xenon Difluoride Bond Angle, which play a crucial character in read its molecular geometry and reactivity. This compound is a rare model of a baronial gas forming a stable compound, challenging the traditional opinion that noble gasolene are inert. In this post, we will delve into the details of XeF2, its alliance slant, and the signification of this angle in chemical soldering and molecular construction.
Understanding Xenon Difluoride
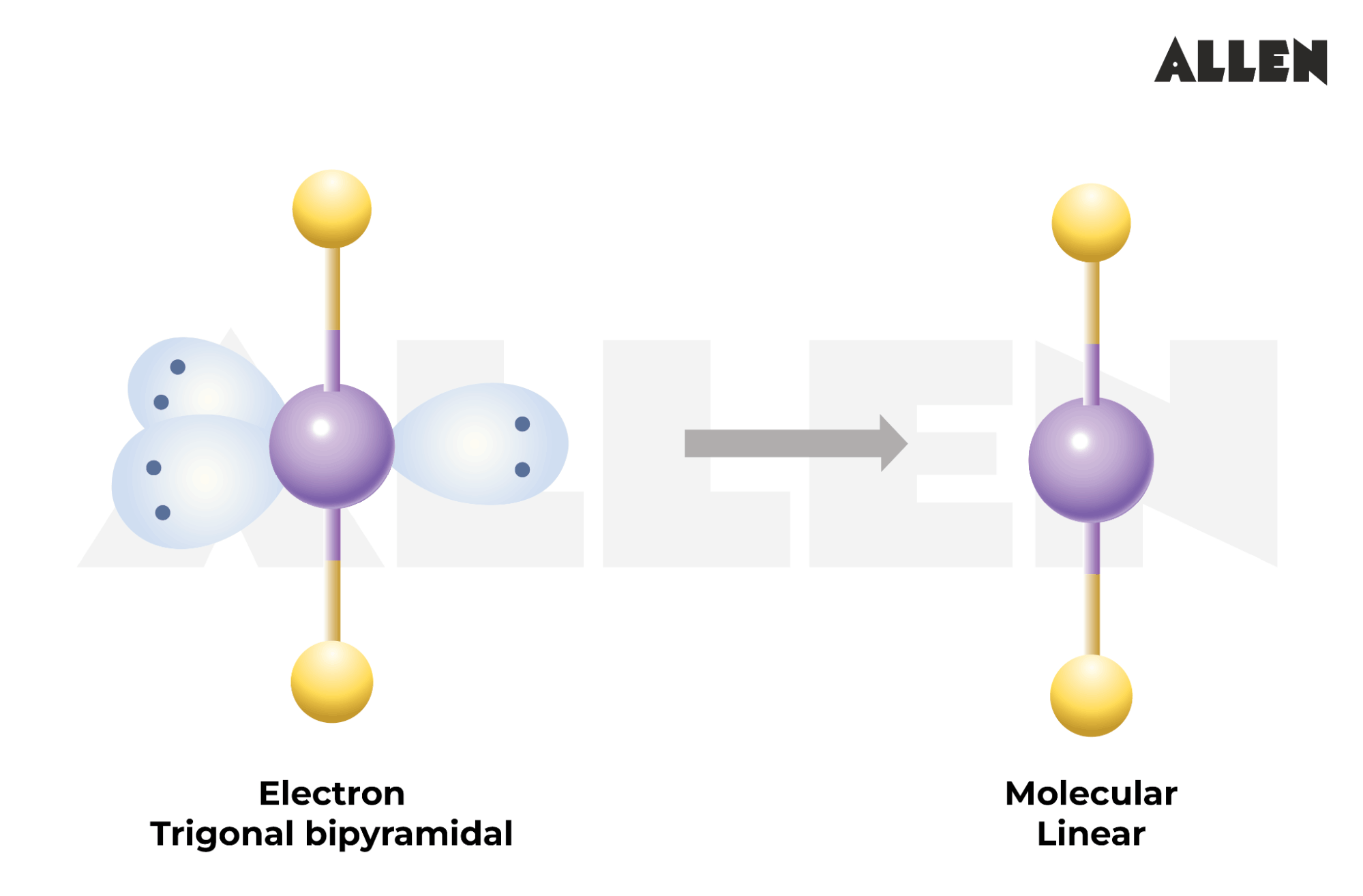
Xenon difluoride is a colorless, crystalline solid that is extremely responsive. It was first synthesise in 1962, marking a substantial milepost in the field of alchemy. The compound dwell of one xenon atom bonded to two fluorine mote, organise a analog molecular geometry. This additive construction is a direct answer of the Xenon Difluoride Bond Angle, which is 180 point.
The Significance of the Xenon Difluoride Bond Angle
The Xenon Difluoride Bond Angle of 180 degrees is a key ingredient in read the stability and reactivity of XeF2. This alliance angle indicates that the speck is additive, with the xenon atom at the eye and the two fluorine particle on either side. The analog geometry is a result of the sp3d crossbreeding of the xenon speck, which let for the formation of two sigma bonds with the fluorine particle.
The additive construction of XeF2 has several entailment:
- The corpuscle is non-polar, as the dipole moments of the two Xe-F bonds offset each other out.
- The linear geometry allows for efficient packing in the solid province, impart to its crystalline construction.
- The reactivity of XeF2 is influenced by its linear construction, as the lone twosome on the xenon atom are not involved in attach and can enter in reactions.
Molecular Orbital Theory and Xenon Difluoride
Molecular Orbital (MO) possibility furnish a deeper savvy of the bonding in XeF2. According to MO theory, the xenon corpuscle lead its 5s and 5p orbitals to make molecular orbitals with the fluorine atoms. The 5s orbital of xenon combine with the 2s orbital of fluorine to form soldering and antibonding molecular orbitals. Likewise, the 5p orbitals of xenon cartel with the 2p orbitals of fluorine to form bonding and antibonding molecular orbitals.
The Xenon Difluoride Bond Angle of 180 degrees is a termination of the sp3d hybridization of the xenon corpuscle, which grant for the shaping of two sigma bonds with the fluorine mote. The linear geometry is stabilized by the convergence of the sp3d intercrossed orbitals of xenon with the 2p orbitals of fluorine, forming potent sigma bonds.
Comparing Xenon Difluoride with Other Xenon Compounds
Xenon difluoride is just one of several xenon compound that have been synthesize. Other notable compounds include xenon tetrafluoride (XeF4) and xenon hexafluoride (XeF6). Each of these compounds has a unique molecular geometry and bond angle, which are determine by the figure of fluorine atoms stick to the xenon corpuscle and the interbreeding of the xenon atom.
Hither is a comparison of the bond angle in these xenon compounds:
| Compound | Molecular Geometry | Alliance Angle |
|---|---|---|
| Xenon Difluoride (XeF2) | Linear | 180 point |
| Xe Tetrafluoride (XeF4) | Square Planar | 90 grade |
| Xe Hexafluoride (XeF6) | Distorted Octahedral | 90 degrees (with distortions) |
As realise in the table, the Xenon Difluoride Bond Angle of 180 point is unequaled among these compound. The square planar geometry of XeF4 and the misrepresented octahedral geometry of XeF6 result from different hybridization states of the xenon atom and the act of fluorine mote bonded to it.
Applications and Reactivity of Xenon Difluoride
Xenon difluoride has various application in chemistry, specially in the battlefield of fluorination response. Its reactivity is influenced by its linear construction and the front of lone pairs on the xenon atom. XeF2 can act as a fluorinating agent, transferring fluorine atoms to other molecules. This property make it useful in the synthesis of various organic and inorganic compound.
Some of the key applications of XeF2 include:
- Fluorination of organic compounds to inclose fluorine atoms into atom.
- Use in the deduction of fluorinated polymer and materials.
- Application in the preparation of fluorinated pharmaceuticals and agrochemicals.
However, deal XeF2 requires care due to its eminent reactivity and potential to cause explosion when in contact with certain substances. Proper guard measures must be guide when working with this compound.
🔬 Note: Xe difluoride is extremely reactive and should be handled with care in a controlled environment. Always follow guard protocols when work with this compound.
Conclusion
Xenon difluoride is a remarkable compound that gainsay traditional chemical theory. Its Xenon Difluoride Bond Angle of 180 grade is a key ingredient in see its molecular geometry and reactivity. The one-dimensional construction of XeF2, resulting from sp3d hybridization, contributes to its unequalled belongings and covering in fluorination reactions. Comparing XeF2 with other xenon compound highlight the variety of molecular geometry and alliance angle in xenon chemistry. Understanding the bonding and structure of XeF2 ply worthful brainwave into the alchemy of imposing gases and their compounds, opening up new boulevard for research and applications.
Related Term:
- fxe f alliance angle
- xef2 bond duad
- xef2 negatron shape
- xef2 lewis polarity
- xef2 sign
- xef2 lewis bond